
The likelihood of a disorderly state versus an orderly state isn't something like 10 to 1 or 3,720 to1, it's more like trillions upon trillions upon trillions (and throw a few more trillions in there just for good measure) to 1.Īs in the case of our once-tidy room, there are very few ways to make a clean room, and an overwhelming number of ways to make a messy one. Ultimately, systems never move from disorder to order (unless there's a way to add energy to them, but that's a story for another article) because of the sheer overwhelming statistics preventing it. And all because there are so, so many more ways for the air to be spread out than crammed into a corner.

These innumerable motions will essentially always leave the air in a disorganized, messy state: evenly spread all throughout the room.
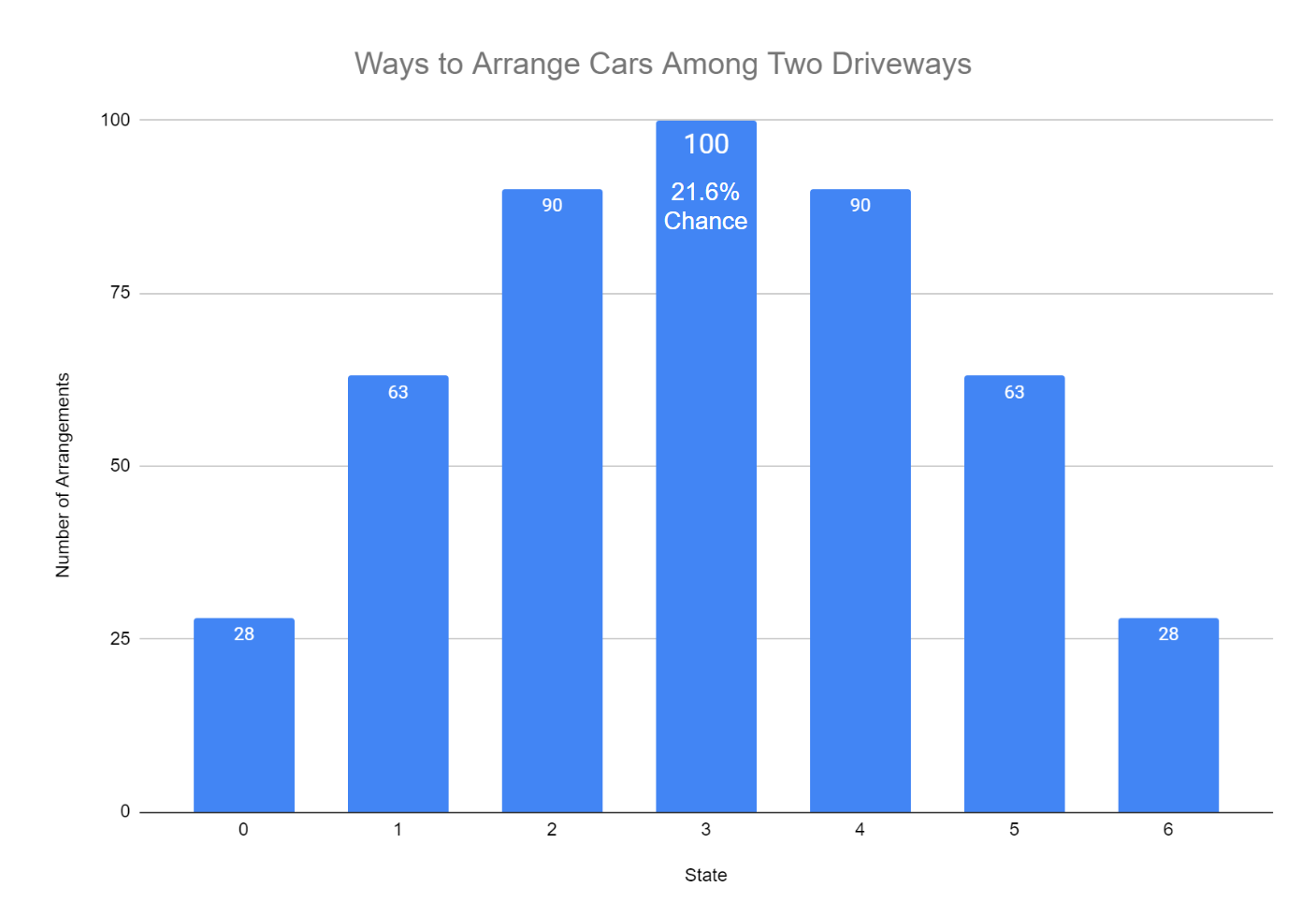
But the motion of air molecules is governed by countless random collisions and movements - a never-ending molecular tornado of activity. Seriously, no law of particle or molecular interactions prevents that. Similarly, there's absolutely nothing stopping the air molecules in your room from collectively deciding to head in the same direction and crowd into the corner, leaving you to asphyxiate in the vacuum. You are much, much more likely to find a messy room after the tornado strike, simply because there are so many more ways for a room to be messy. But that one tiny chance in the ultimate Powerball ticket - and let's face it, you're not going to be the winner. In this example, there are only select arrangements that lead to the measurement of "clean," but there are millions upon millions of possible arrangements that lead to the measurement of "messy." If a tornado were to strike your newly cleaned room, what are the chances of it remaining in the clean state? It's not zero: The tornado, by pure random chance, could pick up every single thing in your room and return it to its original spot. A box of snack crackers on the nightstand? I won't judge, but also messy. How long do you think that Platonic ideal of a room will last? Not long, you realize, as the Sisyphean nature of your endeavors - and perhaps your entire existence - clicks into sharp focus.īut why shouldn't your room stay neat and tidy for years to come? Because if just one thing - one single thing - changes, it's no longer clean.
A place for everything and everything in its place. Imagine finally getting it together, clearing your weekend schedule, waking up early, grabbing that cup of joe, and doing the thing you've been putting off since the holidays: cleaning your room. That's nice, but why should that number never, ever decrease? To explore, let's clean your room. The concept of entropy captures the number of different ways you can rearrange the stuff you can't see (the tiny air particles) to still get the exact same measurements you can see (like the pressure). The pressure, temperature and volume could all remain unchanged.Īnd here's where entropy comes in. Swap one particle with another - would you even notice? Turn a few of the them around - did you even catch it? No, you didn't. There are so many different ways of arranging the air molecules in your box that lead to the exact same pressure, temperature and volume.
